24 January 2024
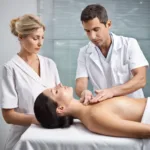
The booming med spa industry is under scrutiny as reports of unlicensed workers, unsanitary conditions, and drug-resistant infections emerge.
Bea Amma’s visit to a med spa in 2021 turned into a nightmare when she contracted a drug-resistant infection that left her scarred and still recovering after more than two years. The med spa industry, which has seen exponential growth in recent years, is now facing scrutiny for its lack of regulation and oversight. With a market value of $15 billion, med spas offer a range of services, from IV therapy to cosmetic procedures, attracting customers with promises of wellness and beauty. However, the lack of standardized regulations and unlicensed workers performing procedures has led to an increase in adverse reactions and infections.
Rise of Med Spas and Lack of Oversight
The med spa industry has experienced a rapid expansion, becoming a multi-billion dollar industry. These facilities offer a wide range of services, including IV therapy, skin care, and cosmetic procedures. However, with this growth comes a lack of oversight and regulation. Federal health officials and representatives of med spa owners have warned that some facilities are using unlicensed workers to inject unapproved products in unsanitary conditions. The absence of federal health regulations and standard operating procedures for med spas leaves the responsibility of oversight to individual states, resulting in inconsistent enforcement.
Increase in Adverse Reactions and Infections
The lack of regulation and oversight in the med spa industry has led to a rise in adverse reactions and infections. Infectious disease and emergency room doctors report seeing more cases associated with med spa procedures. Infections often go unreported to local or state health departments, making it difficult to determine the exact number of people affected. In Bea Amma’s case, she was infected with the aggressive bacterium Mycobacterium abscessus, which is commonly found in water, soil, and dust. Improper sterilization of equipment used in cosmetic procedures can lead to infections.
Unapproved Procedures and Unauthorized Products
The FDA has issued warnings about unapproved procedures and unauthorized products being used in med spas. Consumers have reported severe infections and skin deformities after receiving unauthorized fat-dissolving shots. These shots, sold under various names online, are not approved by the FDA. Some med spas and IV clinics have been found to mix products inappropriately without proper sterilization, leading to serious patient illnesses. The only FDA-approved product for fat reduction is Kybella, which is typically injected under the chin to eliminate a double chin.
Lack of Standards and Oversight
The lack of nationwide standards and consistent oversight contributes to the risks associated with med spas. Currently, there are no federal health regulations or standard operating procedures for these facilities. Each state has its own regulations, but enforcement is often lacking due to resource constraints. The American Med Spa Association aims to fill this gap by providing legal and business resources to med spas across the country. However, unauthorized procedures and unlicensed workers remain a concern in the industry.
Tragic Consequences and Calls for Change
Tragic incidents have occurred at med spas, highlighting the need for increased oversight and regulation. Jenifer Cleveland, who received an IV infusion at a med spa in Texas, passed away shortly after the treatment. The Texas Medical Board suspended the establishment’s medical director’s license for failing to properly supervise an unlicensed individual performing intravenous treatments. Cleveland’s family is calling for increased oversight to prevent similar incidents from happening in the future. The American Med Spa Association also advocates for nationwide standards of practice to ensure consistency and safety.
Conclusion:
The med spa industry’s rapid growth has brought both benefits and risks. While many med spas provide safe and effective treatments, the lack of regulation and oversight has led to a rise in adverse reactions and infections. Consumers are urged to ask questions about the ownership, credentials of practitioners, and presence of licensed medical professionals at med spas. Increased nationwide standards and consistent oversight are necessary to ensure the safety and well-being of individuals seeking treatments at these facilities. As Bea Amma continues her recovery from a drug-resistant infection, the long-lasting impact serves as a reminder of the importance of safety and regulation in the med spa industry.